PBPK for
Radiopharmacology
The use of radiopharmaceuticals dates back to the 1920s, and the field has since grown into a cornerstone of modern diagnostic and therapeutic medicine. Physiologically based pharmacokinetic (PBPK) modeling, first conceptualized in the late 1930s, has become a powerful tool for predicting how compounds move through the body. While PBPK has been applied in various areas of drug development for decades, its integration into radiopharmaceutical research is relatively new, gaining momentum only in the 2010s. Today, ESQlabs is advancing this frontier with its dedicated Radiopharmacology Platform, driving innovation at the intersection of pharmacometrics and nuclear medicine.
Radiopharmacology focuses on radiopharmaceuticals – compounds that pair a biologically active molecule with a radionuclide. These agents are widely used as radiotracers in imaging (e.g., PET and SPECT) and as targeted radiotherapeutics, most often in oncology, where they deliver cytotoxic radiation directly to tumors.
PBPK modeling enhances the development and clinical application of these compounds by simulating their distribution in the body.
Models integrate physiological parameters such as blood flow, organ size, and tissue composition with compound-specific properties including protein binding, lipophilicity, and radionuclide decay.
One of the most promising applications of PBPK in radiopharmacology is its potential to reduce reliance on invasive procedures. By predicting radiotracer behavior more accurately, PBPK can guide the selection of imaging agents and optimize dosing strategies without repeated biopsies or exploratory tests. In therapy, model-informed approaches help maximize delivery of radiation to tumors while minimizing exposure to healthy tissue – improving both safety and effectiveness.
Ultimately, the combination of PBPK modeling and radiopharmaceutical research supports the development of safer, more personalized diagnostic and treatment strategies. These innovations not only advance scientific understanding but also lead to improved patient outcomes, as therapies become more precise, less invasive, and tailored to individual needs.
What we can offer
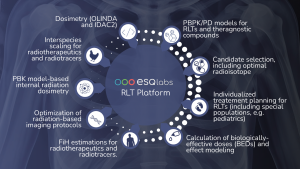
Through our Radiopharmacology Platform, ESQlabs develops and applies PBPK models for both radiotracers and radiotherapeutics. These models support:
Our expertise bridges drug development and clinical application, helping to refine diagnostic and therapeutic protocols while accelerating innovation in radiopharmacology.
Related publications and initatives
Meet the Team

Luis David Jiménez Franco
Luis David Jiménez Franco is an electronics engineer and medical physicist passionate about applying technical, physical and physiological knowledge to improve people’s quality of life. Before joining ESQlabs, he co-developed several physiologically-based pharmacokinetic (PBPK) models for radiopharmaceuticals as well as a PBPK-based algorithm that allows for individualised treatment planning in molecular radiotherapy.
He is experienced in clinical and pre-clinical trials for radiopharmaceuticals and has extensive knowledge of medical imaging and internal radiation dosimetry.
Luis studied Electronics Engineering at the Universidad Pontificia Bolivariana (Colombia) and completed Master’s degrees in Engineering (Universidad EAFIT, Colombia) and Medical Physics (University of Heidelberg, Germany). He received his Ph.D. in Medical Physics under the supervision of Prof. Gerhard Glatting.