Large Molecules,
Biologics and Novel Modalities
Biologics, distinct from traditional small molecules, are advanced medicines based on macro-biomolecules or whole cells and tissues. Typical biologics include antibodies, recombinant proteins, antibody-drug conjugates (ADC), RNA-based vaccines, cell therapies, hormones, etc. The production, assessment, and conduction of trials with biologics require advanced mechanistic models, quality control systems, and risk assessment. Moreover, the high target specificity of most biologics often limits the cross-reactivity in preclinical species, which decreases the translatability of preclinical studies. This increases the need for a mechanistic prediction of the drug effect in humans, based on in vitro data, preclinical data and prior knowledge of the physiology and the therapeutic modality.
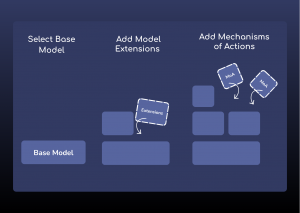
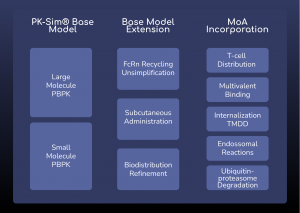
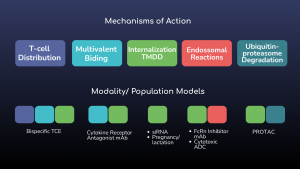
Using modeling approaches, ESQlabs assists in the development and regulatory filing of biological therapeutic solutions. Our modular way of working allows the efficient recombination of specific mechanisms of action with our base PBPK models to create customized models for specific modalities.
Physiologically based biopharmaceutics modeling (PBBM) is a specific field of PBPK model applications that aims to establish the link between the formulation’s properties and in vivo performance.
This field of application of PBPK modeling is evolving at a fast-pace and offers the link between in vivo and in vitro to support pharmaceutical development in the selection of the best drug substance and product, as well as later in development in the establishment of manufacturing quality and controls.
Dissolution testing is often a key input in PBBM. Results from in vitro experiments characterizing drug substances and the formulation behavior (e.g., solubility, particle size, dissolution) can be linked to key ADME parameters and integrated into full PBPK models to predict PK exposure in plasma and/or specific tissues or organs.
These models can also be linked to Pharmacodynamic (PD) relationships to derive the impact of physicochemical, drug and formulation properties on safety and efficacy. The role of Physiologically Based Biopharmaceutics Modeling (PBBM) in drug development spans multiple stages, including supporting patient-centric design, guiding life cycle management, informing regulatory submissions, streamlining development processes, optimizing dosing strategies, enhancing study design, and aiding in formulation development and developability assessment.
Figure 1: ESQlabs modular workflow of model building for large molecule modalities using the latest OSPSuite version 12.
What we can offer
At ESQlabs, we leverage the OSPSuite to deliver customized solutions tailored to your R&D needs. From early discovery to clinical development, we support diverse applications and provide predictive insights to guide decision-making.
Types of prediction
Types of application
Related publications and initatives
Meet the Team

Alexander Kulesza
Alexander is a Chemist by training with a PhD focusing on theoretical and computational methods for structural and optical property predictions.
After spending several years in academia (U. of Lyon) working on molecular dynamics simulation and free energy methods, Alex has most recently been working with CROs in applying large-scale disease and quantitative systems pharmacology models integrated into clinical trial simulations, across a number of disease areas.
Alex leads QSP/T and qAOP / Systems Pharmacology with the aim to promote widespread application of physiologically based and mechanistic modeling and to create robust and qualified, yet versatile models and applications for high impact decision making.

Carla Troisi
Carla joined ESQlabs in early 2024 as part of our large molecules team, bringing her passion for turning complex data into meaningful insights for patient care. While her current focus is on large molecules, she has also built PBPK models for small molecules.
Carla earned her Ph.D. from the University of Bologna, where she specialized in optimizing antibiotic treatments for intensive care patients through the analysis of real-world data. Her research centered on personalizing therapies using a PK/PD approach, particularly by modeling biomarker kinetics like C-reactive protein (CRP).
She further honed her expertise during an internship at Sanofi, where she explored between-species scaling techniques to predict first-in-human doses of monoclonal antibodies using PBPK methodologies.
Carla is also an active member of the GMP association (French group for metabolism and PK), where she has been deeply involved over the years — from student representative to scientific committee member and now board member.

Luis David Jiménez Franco
Luis David Jiménez Franco is an electronics engineer and medical physicist passionate about applying technical, physical and physiological knowledge to improve people’s quality of life. Before joining ESQlabs, he co-developed several physiologically-based pharmacokinetic (PBPK) models for radiopharmaceuticals as well as a PBPK-based algorithm that allows for individualised treatment planning in molecular radiotherapy.
He is experienced in clinical and pre-clinical trials for radiopharmaceuticals and has extensive knowledge of medical imaging and internal radiation dosimetry.
Luis studied Electronics Engineering at the Universidad Pontificia Bolivariana (Colombia) and completed Master’s degrees in Engineering (Universidad EAFIT, Colombia) and Medical Physics (University of Heidelberg, Germany). He received his Ph.D. in Medical Physics under the supervision of Prof. Gerhard Glatting.

Raphaëlle Lesage
Raphaëlle, is a Systems Pharmacologist and Bioengineer by training. At ESQlabs, she focuses on applying PBPK and QSP modelling to support drug development, she coordinates the Special Population core service and is particularly interested in modelling special populations to optimize therapeutic strategies, design trials, and prevent unsafe exposure..
Previously, she served as Chief Scientific Officer at the Virtual Physiological Human Institute, where she coordinated scientific working groups in multiple European projects and led stakeholder engagement and regulatory initiatives for advancing in silico medicine.
She studied Bioengineering and Computational modelling for Biology and Pharmacology at Polytech Nice Sophia. She obtained a degree in reseach valorization from UMPC (Paris) and she holds an interdisciplinary PhD in Engineering and Biomedical Sciences from KU Leuven (Belgium), where she conducted research on computational modelling of therapeutic strategies to limit cartilage degeneration or promote bone regeneration.

Sophie Fischer-Holzhausen
Sophie is a biophysicist dedicated to unraveling the complex interactions underlying physiological processes through mathematical modeling and simulation. She joined ESQlabs in early 2024 as a scientist systems pharmacology.
She earned her Master’s degree in Biophysics from Humboldt University of Berlin, Germany. For her PhD, Sophie joined Prof. Susanna Röblitz’s Computational Systems Biology group at the University of Bergen, Norway, where she helped develop a mechanistic model of menstrual cycle’s endocrine regulation. Prior to joining ESQlabs, she worked as a Pharmacometrician at AstraZeneca in Gothenburg, Sweden.
Sophie is especially passionate about women’s health and leads related initiatives at ESQlabs.

Tatiana Zasedateleva
Tatiana Zasedateleva is a scientist who started her career at ESQlabs conducting research on “Rate-limiting step analysis for target binding in tissue” under the supervision of Dr. Wilbert de Witte. Currently, she actively contributes to projects focusing on the PBPK-modeling of large molecules.
Tatiana holds a degree in Pharmaceutical Sciences from Moscow State University (Russia) and gained diverse professional experience during her academic journey. Her background encompasses research in atherosclerosis, specifically delving into the potential mechanisms of atherothrombogenicity associated with lipoprotein(a). She also was involved in the development of quality control methods for the original drug during her thesis work. In addition to her academic pursuits, Tatiana gained industry experience at Abbott Laboratories, where she worked in the QA&RA department, specializing in medical devices.

Wilbert de Witte
Wilbert de Witte is a Pharmacologist with a strong drive to understand complex mechanisms and the models that represent them. At ESQlabs, he leads the large molecule PBPK and novel modalities platform and he leads the large molecule focus group of the OSP community.
Before joining ESQlabs, he worked at Ablynx NV, later Sanofi Ghent, on the preclinical and clinical development of NANOBODY® therapeutics. He developed several PBPK and PBPK-QSP models as well as traditional TMDD and PKPD models for mechanistic analysis of in vitro, in vivo, and clinical data. He accumulated in-depth knowledge on the behavior of large molecules in different modalities and with various target binding characteristics.
Wilbert de Witte obtained his Master’s degree in Bio-Pharmaceutical Sciences from Leiden University (the Netherlands). For his PhD thesis, he studied the impact of drug-target binding kinetics on in vivo drug action. His PhD research was supervised by Prof. Liesbeth de Lange, Prof. Piet-Hein van der Graaf and Prof. Meindert Danhof at the department of Pharmacology at Leiden University.
- Local depletion of large molecule drugs due to target binding in tissue interstitial space
- Harnessing Open-Source Solutions: Insights From the FirstOpen Systems Pharmacology (OSP) Community Conference
- Harnessing Open-Source Solutions: Insights From the First Open Systems Pharmacology (OSP) Community Conference
- Local depletion of large molecule drugs due to target binding in tissue interstitial space
- Whole-Body Physiologically Based Pharmacokinetic Modeling Framework for Tissue Target Engagement of CD3 Bispecific Antibodies