ADME Characterization:
Data-Poor and
New Chemicals
ESQlabs specializes in characterizing the ADME (absorption, distribution, metabolism, excretion) properties of data-poor or newly developed chemicals and can guide you through the data generation and study design from start to finish.
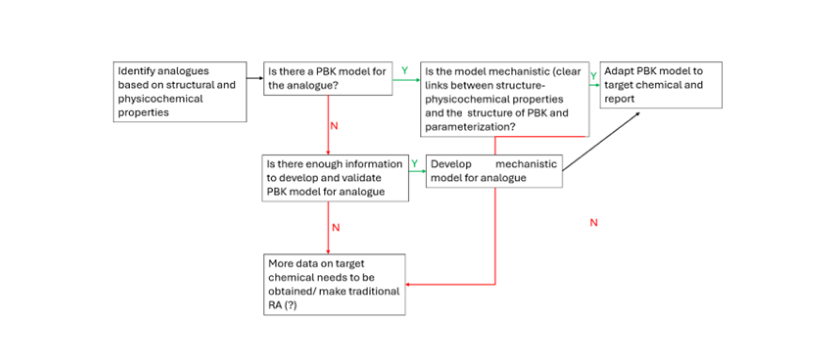
We combine structural similarity analysis, in silico and in vitro ADME kinetics, and mechanistic PBK modelling to develop tailored PBK models for compounds lacking sufficient in vivo TK data, using Read-across approaches. Our methods bridge data gaps, support animal-free assessments, and strengthen regulatory confidence, advancing Next-Generation Risk Assessment (NGRA) goals.
By leveraging read-across, toxicokinetic behavior can be inferred without extensive in vivo testing, aiding risk assessment and safety profiling.
Physiologically Based Kinetic (PBK) models enhance this process by mechanistically describing ADME processes, allowing for quantitative extrapolation across species, exposure routes, and even formulation types.
When combined with structural and metabolic similarity analyses, PBK-based read-across strengthens scientific justification and improves regulatory confidence. Read-across accelerates evaluations, supports Next Generation Risk Assessment (NGRA) goals, and enhances human relevance in toxicology by reducing animal testing.
Figure adapted from Paini, A., Worth, A., Kulkarni, S., Ebbrell, D., Madden, J., 2021.
Assessment of the predictive capacity of a physiologically based kinetic model using a read-across approach.
Computational Toxicology 18, 100159. RA: read-across.
https://doi.org/10.1016/j.comtox.2021.100159
What we can offer
- IVIVE: In vitro–in vivo extrapolation. Translate in vitro ADME data into in vivo toxicokinetic predictions for integration into PBK models, supporting animal-free chemical characterisation.
- High-throughput-PBK · Screening: Rapid, automated PBK parameterisation across large chemical libraries using in silico and in vitro inputs, enabling efficient screening of data-poor or novel compounds.
- Read-Across: Predict ADME behaviour of untested chemicals from structurally similar analogues. PBK models provide quantitative extrapolation even when target-specific data are scarce.
- In Silico ADME: Apply QSAR and PBK parameterisation tools to predict physicochemical and ADME properties without in vivo data.
- ADME Study Design: Expert guidance to design fit-for-purpose ADME studies, identifying data gaps, prioritising experiments, and optimising strategies to generate relevant data efficiently.
Related Platforms
High-Throughput Screening and ADMET-PBPK
Large Molecules, Biologics and Novel Modalities
Small Molecules and Chemicals
Related publications and initatives
Meet the Team

Lara Lamon
Lara Lamon is an Environmental Scientist with a solid drive to investigate the unknowns of chemical exposure and enhance model simulations aimed at protecting human health and the environment.
She worked at ECVAM within the JRC on the grouping and read-across of nanomaterials. Lara contribiuted to applying the read-across framework released by ECHA to support nanomaterials dossiers submissions under the REACH regulation. She further advanced this expertise in the H2020 GRACIOUS project. Additionally, she has developed and applied modeling approaches to estimate environmental emissions and concentrations of environmental and emerging pollutants, including uncertainty analysis (Monte Carlo and sensitivity analysis).
Lara obtained her Ph.D. in Environmental Science at the Ca’ Foscari University of Venice at the Environmental Chemistry and Risk Assessment Unit. During her Ph.D., she was a visiting scholar at the Safety and Environmental Technology group at ETH Zürich, where she collaborated with Martin Scheringer and Matthew Macleod at the Safety and Technology group.

Marco Siccardi
Marco is a Clinical Biologist by training with a PhD in molecular pharmacology and PK/PD modelling. He spent over 15 years at the University of Liverpool working on the topic of pharmacogenetics and in developing PBPK approaches for the optimisation of drug delivery, including HIV therapy optimisation.
Marco has most recently been working with CROs in taking this approaches for modelling and simulation approaches and PKTK (Systems Toxicology) models across a number of disease areas.
Marco leads the Systems Toxicology team with the aim to promote collaborative innovation and to develop novel modeling approaches to streamline the toxicological assessment.
- Development of an end-to-end Quantitative Model-Informed Drug Development (MIDD) ECOSYSTEM
- A review of OSP suite PBBM capabilities: looking ahead
- Roadmap for action for advancing aggregate exposure to chemicals in the EU
- Application of High-Throughput PBPK Modeling to Develop an IVIVE Approach for Oral Permeability
- Enhancing PB(P)K Models for the Female Reproductive Tract: A Framework for Local and Systemic Drug Kinetics
- Advancing Maternal-Fetal and Lactation PBK Models for Cross-Species Risk Assessment in Toxicology
- High-Throughput PBPK Framework in R using Open Systems Pharmacology Software for Anti-Tuberculosis Drug Development

Marjory Moreau
A leading expert in human systems biology and chemical safety assessment, Marjory brings world-class expertise in physiologically-based pharmacokinetic (PBK) modeling and quantitative in vitro to in vivo extrapolation (QIVIVE). With a PhD in Toxicology from the University of Montreal and a postdoctoral fellowship at Health Canada’s Computational Toxicology Laboratory, she has dedicated her career to advancing 𝐍𝐞𝐱𝐭-𝐆𝐞𝐧𝐞𝐫𝐚𝐭𝐢𝐨𝐧 𝐑𝐢𝐬𝐤 𝐀𝐬𝐬𝐞𝐬𝐬𝐦𝐞𝐧𝐭 (𝐍𝐆𝐑𝐀) and bridging cutting-edge science with 𝐫𝐞𝐠𝐮𝐥𝐚𝐭𝐨𝐫𝐲 𝐝𝐞𝐜𝐢𝐬𝐢𝐨𝐧-𝐦𝐚𝐤𝐢𝐧𝐠. At ESQlabs, she will lead NGRA initiatives in the U.S., positioning us as a trusted regulatory partner while strengthening support for our American clients.

Stella Fragki
Styliani (Stella) Fragki is a Senior Scientist in Systems Toxicology at ESQlabs, where she applies physiologically based kinetic (PBK) modeling in chemical risk assessment. Her work focuses on integrating New Approach Methodologies (NAMs) within the framework of Next-Generation Risk Assessment (NGRA), with the aim of supporting human-relevant and animal-free safety evaluations.
She is a certified European Registered Toxicologist and has been active in the field of toxicological risk assessment since 2010, supporting the (agro)chemical industry with dossier preparation for plant protection products, biocides, and REACH-regulated substances.
Stella holds a degree in Biology from Thessaloniki, Greece, and an MSc in Food Safety from Wageningen University in the Netherlands. Her PhD research focused on quantitative in vitro to in vivo extrapolation (QIVIVE) using physiologically based kinetic (PBK) modeling, integrating data from both in vitro and in silico sources. Stella is also a certified European Registered Toxicologist.
- Towards best use and regulatory acceptance of generic physiologically based kinetic (PBK) models for in vitro-to-in vivo extrapolation (IVIVE) in chemical risk assessment
- Towards best use and regulatory acceptance of generic physiologicallybased kinetic (PBK) models for in vitro‑to‑in vivo extrapolation (IVIVE)in chemical risk assessment
- Advancing PFAS risk assessment Integrative approaches using agent-basedmodelling and physiologically-based kinetic for environmental andhealth safety
- EFSA Project on the use of NAMs to explore the immunotoxicity of PFAS
- Advancing Maternal-Fetal and Lactation PBK Models for Cross-Species Risk Assessment in Toxicology

Susana Proença
Susana Proença is a biologist and toxicologist dedicated to the leveraging in vitro and in silico data for parameterizing PBPK models and to perform chemical safety assessment. She is focused on developing frameworks and case-studies for IVIVE, extrapolation of ADME properties from in vitro to in vivo, and on QIVIVE, extrapolating in vitro effect concentrations to in vivo doses. She has experience in working with PBPK models both in OSP and in R.
Before joining ESQlabs, she worked at Wageningen University, Toxicology division under Dr. Nynke Kramer supervision. There she worked on evaluating in vitro kinetics of chemical related to different toxicological ontologies (such as cholestasis and development neurotoxicity) and developing strategies for performing QIVIVE for these chemicals. Before this she underwent an internship at ECVAM-JRC on in silico modelling of in vitro kinetics, which was followed by a stint automating chemical data curation from REACH dossiers, also in JRC.
Susana obtained her Master’s degree in Bio-Pharmaceutical Sciences from Faculty of Pharmaceutical Sciences, Lisbon University (Portugal). For her PhD thesis, she studied the in vitro kinetics in complex in vitro models and (Q)IVIVE of highly lipophilic chemicals. The thesis was supervised by Dr. Nynke Kramer at the Institute for Risk Assessment Sciences at Utrecht University. The work was multidisciplinary, envolving setting in vitro experiments, analytical methods, transcriptomics analysis and in silico modelling. Her PhD thesis will be submitted soon.
- Workshop Report no.40 – Chronos and Kairos: Understanding time in biology for NGRA
- Application of High-Throughput PBPK Modeling to Develop an IVIVE Approach for Oral Permeability
- Effective exposure of chemicals in in vitro cell systems: A review of chemical distribution models
- Harnessing Open-Source Solutions: Insights From the FirstOpen Systems Pharmacology (OSP) Community Conference
- Harnessing Open-Source Solutions: Insights From the First Open Systems Pharmacology (OSP) Community Conference